Image Details
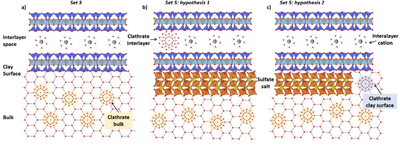
Caption: Figure 4.
Schematic representation of the hypothetical final situations of the systems with montmorillonite. (a) Set 3 H2O–Mt–CH4: where the only melting peak of clathrate is related to those formed in the bulk (Figures A5 and A6), (although we cannot exclude the presence of minor amounts of clathrate formed in the interlayer); (b) Set 5 MgSO4 17wt%–Mt–CH4, hypothesis 1: assuming that the first melting clathrate peak (peak 1) is related to those located in the bulk and the second (peak 2) in the interlayer (Figures A9 and A10); (c) Set 5 MgSO4 17wt%–Mt–CH4, hypothesis 2: assuming that the first clathrate melting peak (peak 1) is related to those located on the clay surface and the second one (peak 2) in the bulk (Figures A9 and A10)
Copyright and Terms & Conditions
© 2026. The Author(s). Published by the American Astronomical Society.